BRUKINSA patients can call the myBeiGene® patient support program to talk to a dedicated nurse: 1-833-BEIGENE (1-833-234-4363)
COHORT 1: OVERALL INCIDENCE OF ADVERSE REACTIONS (ARs)1,2
| Adverse Reactions | ARs in ≥10% of Patients Without Del(17p) | Pooled Safety Population* | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=240) | BR (n=227) | BRUKINSA (N=1550) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Musculoskeletal pain | 33 | 2 | 17 | 0.4 | 30 | 2 |
| Upper respiratory tract infection | 28 | 1 | 15 | 0.9 | 39 | 2 |
| Pneumonia | 13† | 5 | 8‡ | 4 | 20 | 11 |
| Hemorrhage | 27† | 4 | 4 | 0.4 | 30 | 4 |
| Hypertension | 14 | 7 | 5 | 3 | 14 | 7 |
| Rash | 24 | 1 | 30 | 5 | 28 | 0.9 |
| Bruising | 24 | 0 | 3 | 0 | 23 | 0.1 |
| Cough | 15 | 0 | 10 | 0 | 19 | 0.1 |
| Diarrhea | 14 | 0.8 | 12‡ | 0.9 | 19 | 2 |
| Constipation | 10 | 0.4 | 18 | 0 | 13 | 0.3 |
| Nausea | 10 | 0 | 33 | 1 | 11 | 0.2 |
| Fatigue | 14 | 1 | 21 | 2 | 17 | 1 |
| Second primary malignancy | 13† | 6 | 1 | 0.4 | 13 | 6 |
| Headache | 12 | 0 | 8 | 0 | 11 | 0.4 |
| Dizziness | 11 | 0.8 | 5 | 0 | 11 | 0.3 |
*Includes chronic lymphocytic leukemia, Waldenström’s macroglobulinemia, mantle cell lymphoma, follicular lymphoma, marginal zone lymphoma, hairy cell leukemia, diffuse large B-cell lymphoma, and Richter’s transformation.2
†Includes 3 fatal outcomes.1
‡Includes 2 fatal outcomes.1
COHORT 2: OVERALL INCIDENCE OF ARs1,2
| Adverse Reactions | ARs in ≥10% of Patients With Del(17p) | Pooled Safety Population* | ||
|---|---|---|---|---|
| BRUKINSA (n=111) | BRUKINSA (N=1550) | |||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Upper respiratory tract infection | 38 | 0 | 39 | 2 |
| Pneumonia | 20† | 8 | 20 | 11 |
| Musculoskeletal pain | 38 | 3 | 30 | 2 |
| Rash | 28 | 0 | 28 | 0.9 |
| Bruising | 26 | 0.9 | 23 | 0.1 |
| Hemorrhage | 28 | 5 | 30 | 4 |
| Hypertension | 11 | 5 | 14 | 7 |
| Second primary malignancy | 22‡ | 6 | 13 | 6 |
| Diarrhea | 18 | 0.9 | 19 | 2 |
| Nausea | 16 | 0 | 11 | 0.2 |
| Constipation | 15 | 0 | 13 | 0.3 |
| Abdominal pain | 12 | 2 | 10 | 0.6 |
| Cough | 18 | 0 | 19 | 0.1 |
| Dyspnea | 13 | 0 | 8 | 0.5 |
| Fatigue | 14 | 0.9 | 17 | 1 |
| Headache | 11 | 2 | 11 | 0.4 |
*Includes chronic lymphocytic leukemia, Waldenström’s macroglobulinemia, mantle cell lymphoma, follicular lymphoma, marginal zone lymphoma, hairy cell leukemia, diffuse large B-cell lymphoma, and Richter’s transformation.2
†Includes 1 fatal outcome.1
‡Includes non-melanoma skin cancer in 13%.1
| Laboratory Abnormality | Cohort 1: Patients Without Del(17p) | Cohort 2: Patients With Del(17p) | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=239)* | BR (n=227) | BRUKINSA (n=111)† | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Neutrophils decreased | 37 | 15 | 80 | 53 | 42 | 19‡ |
| Hemoglobin decreased | 29 | 3 | 66 | 8 | 26 | 4 |
| Platelets decreased | 27 | 2 | 61 | 11 | 23 | 0.9 |
| Leukocytes increased | 21§ | 21 | 0.4 | 0.4 | NR | NR |
| Glucose increased¶ | 55 | 7 | 67 | 10 | 52 | 6 |
| Creatinine increased | 22 | 0.8 | 18 | 0.4 | 27 | 0.9 |
| Magnesium increased | 22 | 0 | 14 | 0.4 | 31 | 0 |
| Alanine aminotransferase increased | 21 | 2 | 23 | 2 | NR | NR |
*In Cohort 1, the denominator used to calculate the rate varied from 239 to 227 based on the number of patients with a baseline value and at least 1 post-treatment value. Grading is based on NCI CTCAE criteria.
†In Cohort 2, the denominator used to calculate the rate varied from 110 to 111 based on the number of patients with a baseline value and at least 1 post-treatment value. Grading is based on NCI CTCAE criteria.
‡Grade 4, 9%.
§Lymphocytes increased in 15%.
¶Patients on study were not required to fast for lab tests.
| Adverse Events | SEQUOIA | Pooled Safety Population* | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=240) | BR (n=227) | BRUKINSA (N=1550) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Fatigue | 12 | 1 | 15 | 0.9 | 17 | 1 |
| Headache | 11 | 0 | 7 | 0 | 11 | 0.4 |
| Myalgia | 4 | 0 | 1 | 0 | 4 | 0.4 |
| Arthralgia | 13 | 0.8 | 9 | 0.4 | 14 | 0.7 |
| Atrial fibrillation/flutter† | 3 | 0.4 | 3 | 1 | 4 | 2 |
| Hypertension | 14 | 6 | 11 | 5 | 14 | 7 |
| Major bleeding‡ | 5 | 4 | 2 | 2 | 5 | 4 |
*Includes chronic lymphocytic leukemia, Waldenström’s macroglobulinemia, mantle cell lymphoma, follicular lymphoma, marginal zone lymphoma, hairy cell leukemia, diffuse large B-cell lymphoma, and Richter’s transformation.2
†Includes rates of atrial fibrillation only; does not include flutter.2
‡Major bleeding includes subdural hematoma or subdural hemorrhage.
LOWER RATES OF DOSE REDUCTIONS AND DISCONTINUATION DUE TO AEs3
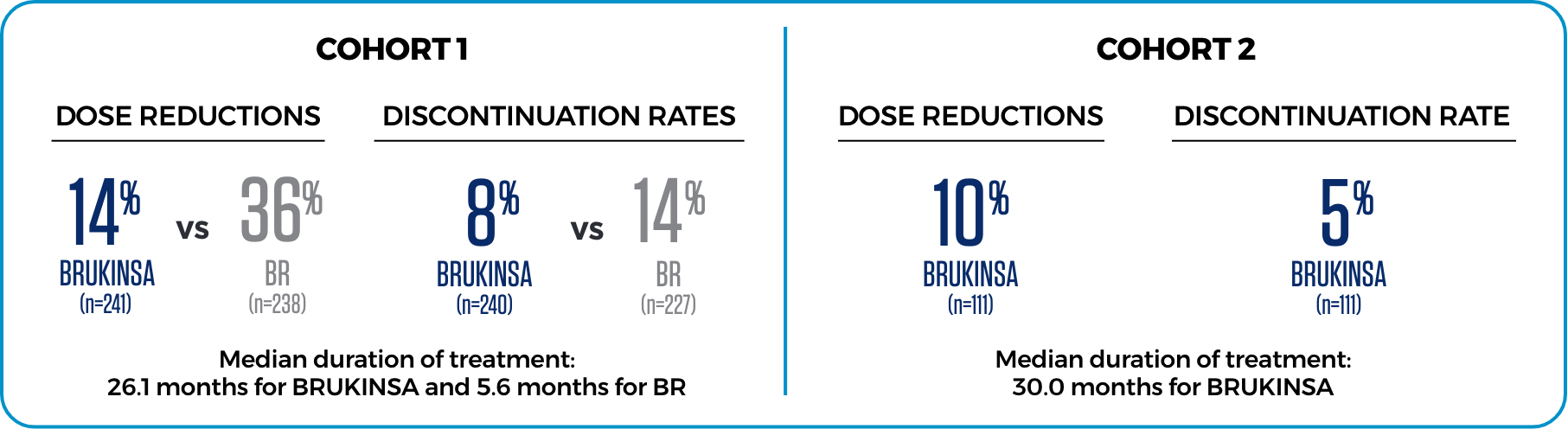
AE=adverse event; AR=adverse reaction; BR=bendamustine+rituximab; NCI CTCAE=National Cancer Institute Common Terminology Criteria for Adverse Events; NR=not reported.
| Adverse Reactions | ARs in ≥10% of Patients | Pooled Safety Population* | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=324) | Ibrutinib (n=324) | BRUKINSA (N=1550) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Upper respiratory tract infection | 27 | 1 | 22 | 1 | 39 | 2 |
| Pneumonia | 18† | 9 | 19‡ | 11 | 20 | 11 |
| COVID-19 | 14† | 7 | 10‡ | 5 | 5 | 3 |
| Musculoskeletal pain | 26 | 0.6 | 28 | 0.6 | 30 | 2 |
| Hemorrhage | 24† | 3 | 26‡ | 4 | 30 | 4 |
| Hypertension | 19 | 13 | 20 | 13 | 14 | 7 |
| Rash | 20 | 1 | 21 | 0.9 | 28 | 0.9 |
| Bruising | 16 | 0 | 14 | 0 | 23 | 0.1 |
| Diarrhea | 14 | 2 | 22 | 0.9 | 19 | 2 |
| Fatigue | 13 | 0.9 | 14 | 0.9 | 17 | 1 |
| Cough | 11 | 0.3 | 11 | 0 | 19 | 0.1 |
| Dizziness | 10 | 0 | 7 | 0 | 11 | 0.3 |
Rates of hypertension were comparable between BRUKINSA and ibrutinib1
- A medical history of hypertension was reported in more than half of these patient events for both BRUKINSA and ibrutinib2
*Includes chronic lymphocytic leukemia, Waldenström’s macroglobulinemia, mantle cell lymphoma, follicular lymphoma, marginal zone lymphoma, hairy cell leukemia, diffuse large B-cell lymphoma, and Richter’s transformation.2
†Includes fatal outcomes: pneumonia (9 patients), COVID-19 (8 patients), and hemorrhage (1 patient).1
‡Includes fatal outcomes: pneumonia (10 patients), COVID-19 (9 patients), and hemorrhage (2 patients).1
| Laboratory Abnormality | BRUKINSA (n=321) | Ibrutinib (n=321)* | ||
|---|---|---|---|---|
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Neutrophils decreased | 43 | 15 | 33 | 16 |
| Hemoglobin decreased | 28 | 4 | 32 | 4 |
| Lymphocytes increased | 24 | 19 | 26 | 19 |
| Platelets decreased | 22 | 4 | 24 | 3 |
| Glucose increased† | 52 | 5 | 29 | 3 |
| Creatinine increased | 26 | 0 | 23 | 0 |
| Phosphate decreased | 21 | 3 | 13 | 2 |
| Calcium decreased | 21 | 0.6 | 29 | 0 |
*The denominator used to calculate rates of lab abnormalities varied from 320 to 321 in the ibrutinib arm based on the number of patients with a baseline value and at least 1 post-treatment value. Grading is based on NCI CTCAE criteria.
†Patients on study were not required to fast for lab tests.
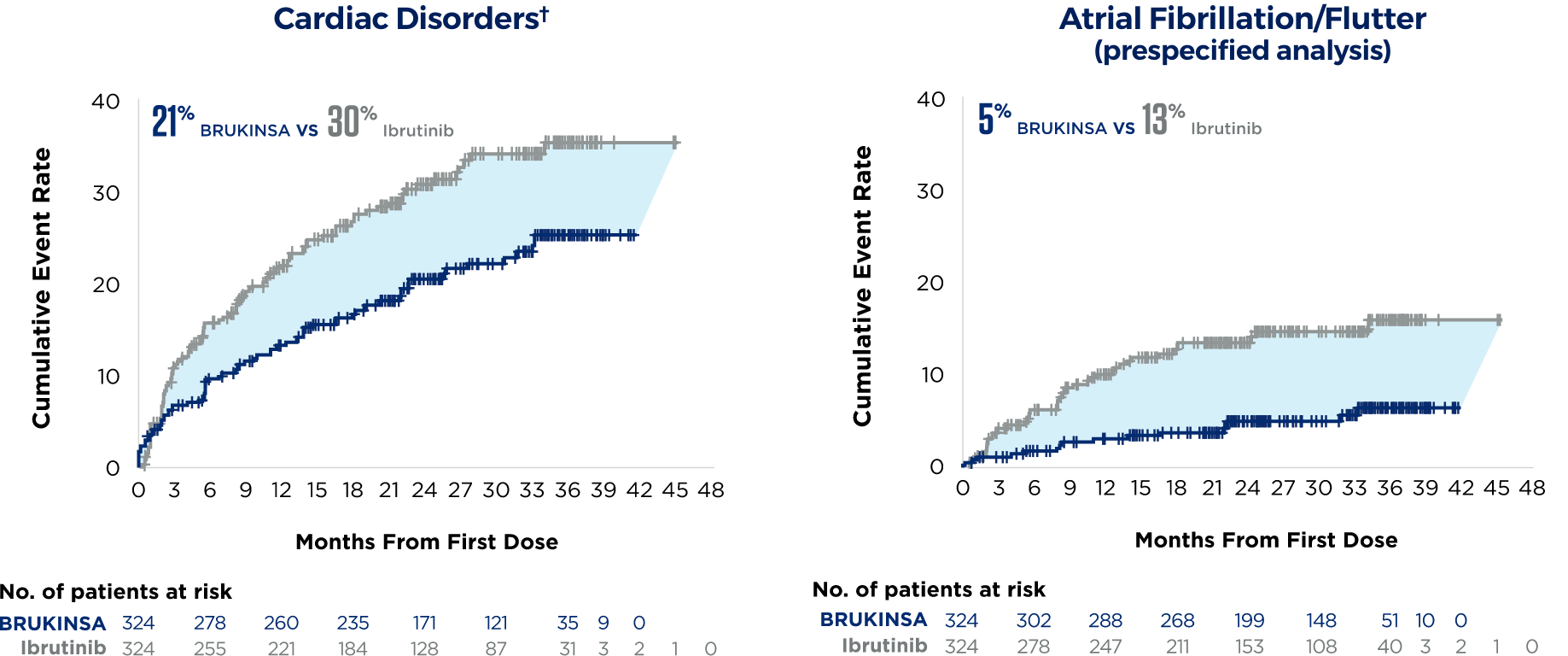
*Assessed by both IRC and investigator with similar results. Median duration of treatment: 28.4 months for BRUKINSA and 24.3 months for ibrutinib.4
†Cardiac disorders is a grouped term that includes atrial fibrillation and atrial flutter.4
| BRUKINSA (n=324) | Ibrutinib (n=324) | |
|---|---|---|
| Cardiac adverse events | 69 (21.3%) | 96 (29.6%) |
| Serious cardiac adverse events | 6 (1.9%) | 25 (7.7%) |
| Cardiac adverse events leading to treatment discontinuation* | 1 (0.3%) | 14 (4.3%) |
| Fatal cardiac events | 0 (0%) | 6 (1.9%) |
*BRUKINSA cardiac-related discontinuation in 1 patient was for ventricular extrasystoles. Ibrutinib cardiac-related discontinuations were for atrial fibrillation (5), cardiac arrest (2), cardiac failure (2), cardiac failure acute (1), congestive cardiomyopathy (1), myocardial infarction (1), palpitations (1), and ventricular fibrillation (1).4
There were 6 fatal cardiac events in patients treated with ibrutinib and none with BRUKINSA4
Assessed by both IRC and investigator with similar results. Median duration of treatment: 28.4 months for BRUKINSA and 24.3 months for ibrutinib.4
| Adverse Reactions | ALPINE | Pooled Safety Population* | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=324) | Ibrutinib (n=324) | BRUKINSA (N=1550) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Fatigue | 13 | 0.9 | 14 | 0.9 | 17 | 1 |
| Headache | 8 | 0 | 9 | 0 | 11 | 0.4 |
| Myalgia | 3 | 0 | 4 | 0 | 4 | 0.4 |
| Arthralgia | 14 | 0 | 15 | 0.3 | 14 | 0.7 |
| Atrial fibrillation and flutter | 5 | 3 | 13 | 4 | 4 | 2 |
| Hypertension | 19 | 13 | 20 | 13 | 14 | 7 |
| Major hemorrhage† | 4 | 3 | 4 | 4 | 5 | 4 |
*Includes chronic lymphocytic leukemia, Waldenström’s macroglobulinemia, mantle cell lymphoma, follicular lymphoma, marginal zone lymphoma, hairy cell leukemia, diffuse large B-cell lymphoma, and Richter’s transformation.2
†Hematuria was the most common major hemorrhage.2

1 patient in the BRUKINSA arm discontinued treatment due to a cardiac AE vs 14 patients in the ibrutinib arm*
*BRUKINSA cardiac-related discontinuation in 1 patient was for ventricular extrasystoles. Ibrutinib cardiac-related discontinuations were for atrial fibrillation (5), cardiac arrest (2), cardiac failure (2), cardiac failure acute (1), congestive cardiomyopathy (1), myocardial infarction (1), palpitations (1), and ventricular fibrillation (1).
AE=adverse event; afib=atrial fibrillation; AR=adverse reaction; IRC=independent review committee; NCI CTCAE=National Cancer Institute Common Terminology Criteria for Adverse Events.
Dr Anthony Nguyen discusses the safety profile of BRUKINSA vs ibrutinib in CLL
DISCOVER MORE VIDEOSA global supportive Phase 1/2, open-label, single-arm trial that included patients with treatment-naïve or relapsed/refractory CLL/SLL and evaluated ~4 years of safety and ~3 years of efficacy data.6
ADVERSE EVENTS OF SPECIAL INTEREST DECREASED OVER TIME6Incidence of Treatment-Emergent Adverse Events (TEAEs) Over Time (Grade ≥3)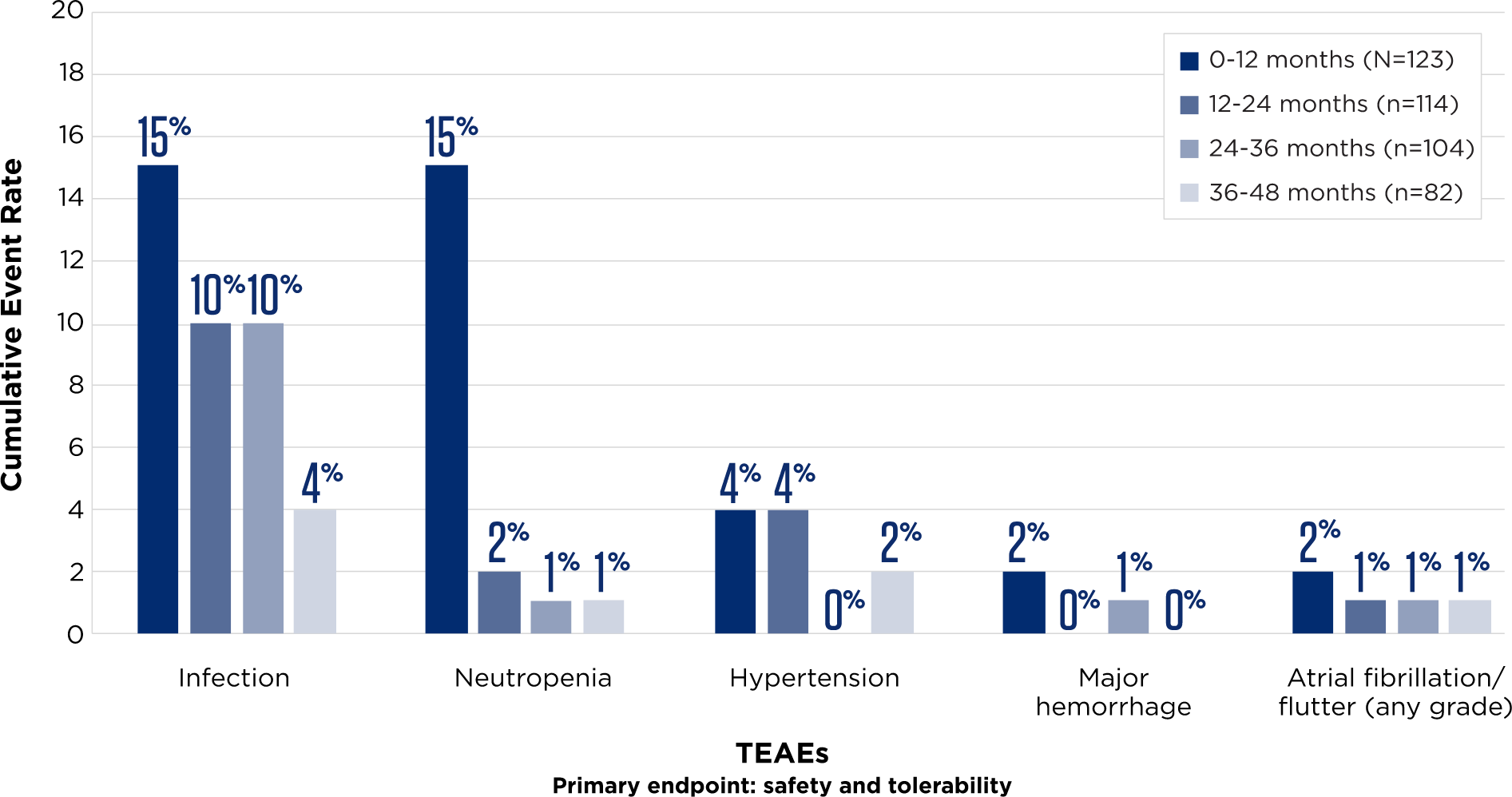
- Atrial fibrillation remained consistently low
- No new safety signals
- Low discontinuation rate of 10% over 4 years (N=123)
TEAEs were consistent with the overall safety profile of BRUKINSA
Overall median duration of treatment: 43 months.
Exploratory long-term analysis; all data are descriptive in nature.
Study 003 was a global supportive Phase 1/2 trial for 1L and 2L CLL.
*Assessed by IRC.
1L=first line; 2L=second line; CLL=chronic lymphocytic leukemia; IRC=independent review committee; SLL=small lymphocytic lymphoma.
IMPORTANT SAFETY INFORMATION
What should I tell my healthcare provider before taking BRUKINSA?
Before taking BRUKINSA, tell your healthcare provider about all of your medical conditions, including if you:
- have bleeding problems.
- have had recent surgery or plan to have surgery. Your healthcare provider may stop BRUKINSA for any planned medical, surgical, or dental procedure.
- have an infection.
- have or had heart rhythm problems.
- have high blood pressure.
- have liver problems, including a history of hepatitis B virus (HBV) infection.
- are pregnant or plan to become pregnant. BRUKINSA can harm your unborn baby. If you are able to become pregnant, your healthcare provider may do a pregnancy test before starting treatment with BRUKINSA.
- Females should avoid getting pregnant during treatment and for 1 week after the last dose of BRUKINSA. You should use effective birth control (contraception) during treatment and for 1 week after the last dose of BRUKINSA.
- Males should avoid getting female partners pregnant during treatment and for 1 week after the last dose of BRUKINSA. You should use effective birth control (contraception) during treatment and for 1 week after the last dose of BRUKINSA.
- are breastfeeding or plan to breastfeed. It is not known if BRUKINSA passes into your breast milk. Do not breastfeed during treatment with BRUKINSA and for 2 weeks after the last dose of BRUKINSA.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking BRUKINSA with certain other medications may affect how BRUKINSA works and can cause side effects.
What are the possible side effects of BRUKINSA?
BRUKINSA may cause serious side effects, including:
- Bleeding problems (hemorrhage). Bleeding problems are common with BRUKINSA, and can be serious and may lead to death. Your risk of bleeding may increase if you are also taking a blood thinner medicine. Tell your healthcare provider if you have any signs or symptoms of bleeding, including:
- blood in your stools or black stools (looks like tar)
- pink or brown urine
- unexpected bleeding, or bleeding that is severe or you cannot control
- vomit blood or vomit that looks like coffee grounds
- cough up blood or blood clots
- increased bruising
- dizziness
- weakness
- confusion
- change in speech
- headache that lasts a long time
- Infections that can be serious and may lead to death. Tell your healthcare provider right away if you have fever, chills, or flu-like symptoms.
- Decrease in blood cell counts (white blood cells, platelets, and red blood cells). Your healthcare provider should do blood tests during treatment with BRUKINSA to check your blood counts.
- Second primary cancers. New cancers have happened in people during treatment with BRUKINSA, including cancers of the skin or other organs. Your healthcare provider will check you for other cancers during treatment with BRUKINSA. Use sun protection when you are outside in sunlight.
- Heart rhythm problems (atrial fibrillation, atrial flutter, and ventricular arrhythmias) that can be serious and may lead to death. Tell your healthcare provider if you have any of the following signs or symptoms:
- your heartbeat is fast or irregular
- feel lightheaded or dizzy
- pass out (faint)
- shortness of breath
- chest discomfort
The most common side effects of BRUKINSA include:
- decreased white blood cell count
- decreased platelet count
- upper respiratory tract infection
- bleeding
- muscle, bone, or joint pain
These are not all the possible side effects of BRUKINSA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
What is BRUKINSA?
BRUKINSA is a prescription medicine used to treat adults with:
- Chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL).
- Waldenström’s macroglobulinemia (WM).
- Mantle cell lymphoma (MCL) who have received at least one prior treatment for their cancer.
- Marginal zone lymphoma (MZL) when the disease has come back or did not respond to treatment and who have received at least one certain type of treatment.
- Follicular lymphoma (FL), in combination with the medicine obinutuzumab, when the disease has come back or did not respond to treatment and who have received at least two prior treatments.
It is not known if BRUKINSA is safe and effective in children.
Please see full Prescribing Information including Patient Information.

